
I had been thinking of setting up an aquarium for a long time, so one fine day I ordered one at the local fish store. The size is 160 x 60 x 60 cm = 576 liters. It was delivered to the shop, so I had to hire a moving company to get it up to my apartment. While they were at it, they could just as well carry all the other stuff (gravel, decoration etc) as well, leaving me to just receive the goods at my doorstep.
Setting up a new fish tank is not quite as easy as it might look like. It's easy to set up the tank with decoration and water and put in some plants and fish, and it all looks very well for some time. Then all the problems there may be begin to show. Plants start to turn yellow or just don't grow, and algae and snails appear.
An experienced aquarium owner knows just what to do in such situations. Others - like me - read a lot of pages on the Internet about it, and here is my page about aquarium maintenance and care.
Setting up a new fish tank is not quite as easy as it might look like. It's easy to set up the tank with decoration and water and put in some plants and fish, and it all looks very well for some time. Then all the problems there may be begin to show. Plants start to turn yellow or just don't grow, and algae and snails appear.
An experienced aquarium owner knows just what to do in such situations. Others - like me - read a lot of pages on the Internet about it, and here is my page about aquarium maintenance and care.
A new aquarium

An aquarium can be "low-tech" or "hi-tech".
Low-tech aquarium
A low-tech aquarium has only the minimum equipment and care. The low-tech is without CO2 and fertilizer and with lower lighting to compensate for the lower CO2 and fertilizer levels.
High-tech aquarium
Hi-tech is with everything: Extra CO2 and fertilizer and reverse-osmosis water and high-quality substrate and high lighting to help the plants consume all this added CO2 and fertilizer. Some fish and plants only thrive in hi-tech aquariums.
There is a huge concept called water chemistry. An aquarium does not need to be hi-tech to have a good water chemistry, is's all about the balance of various elements in the water.
Low-tech aquarium
A low-tech aquarium has only the minimum equipment and care. The low-tech is without CO2 and fertilizer and with lower lighting to compensate for the lower CO2 and fertilizer levels.
High-tech aquarium
Hi-tech is with everything: Extra CO2 and fertilizer and reverse-osmosis water and high-quality substrate and high lighting to help the plants consume all this added CO2 and fertilizer. Some fish and plants only thrive in hi-tech aquariums.
There is a huge concept called water chemistry. An aquarium does not need to be hi-tech to have a good water chemistry, is's all about the balance of various elements in the water.
Equipment
|
External filter
|
Highly recommended for all tanks. External filters are more easy to clean than internal filter pumps and external filter pumps don't take up space in the tank. The filter pump provides the essential low circulation in the tank to prevent stillwater pockets.
|
|
Powerheads and circulation pumps
|
Recommended for larger tanks to ensure that the water is circulated properly in the entire tank. It's easy to put in a too large pump. Although surface agitation is generally a good thing, it can remove the added CO2 from the water.
|
|
Heaters
|
Make sure that the fish and plants in the aquarium have compatible temperature requirements.
|
|
Light
|
A high-tech aquarium requires more light than a low-tech aquarium. Too many hours of light per day cause algae attack.
|
|
CO2
|
CO2 is added in hi-tech aquariums. CO2 is necessary for the photosynthesis process, and if there isn't enough, the plants won't grow. There are a number of CO2-sytems available, from DIY to pressure-bottle systems with a regulator and bubble-counter and a reactor/diffuser to dissolve the CO2 in the water.
|
|
Reverse osmosis
|
Reverse osmosis (abbreviated RO) is a method of purifying the tap water. Tap water contains minerals and other additives, this can be filtered out in a RO system, giving "ultra-pure" and soft water for the aquarium. There are many reasons to prefer RO water over normal tap water, but RO kits are expensive and large and produce purified water at a very slow rate.
|
|
Fertilizer
|
Fertilizer is for plants what fish food is for fish. Needed in hi-tech aquariums, recommended for low-tech aquariums except for purists.
|
|
Substrate
|
The cheapest substrate is just crushed rock or gravel more or less course or fine. It even exists in various colors. More (very much more) expensive types of substrate exist, and they all promise improved conditions for plant growth. This is also true, more true for difficult plants, easy plants will grow fine in cheap substrate. The more expensive substrate allows for thinner plant roots and can hold on to nutrients better than a simple rock surface can. Difficult plants will have a hard time growing in the cheap crushed rock substrate.
|
|
Substrate additives
|
When using an advanced type of substrate, then why not improve it even further by boosting the eco-system with some presets? Recommended for hi-tech purists, but not strictly necessary. A freshly setup aquarium has no eco-system except what has just been put into it, but it develops over time. When presets are added, some large variations in water chemistry can occur in the first few weeks, which can be dangerous to fish. Read the instructions on the product.
|

Winning the competition against algae
Every aquarium has a small algae population, and that's fine, as long as the algea don't start taking over the entire aquarium.
One way to control algae is to have fish or shrimps eating them. The Siamensis Flying Fox is an excellent algae eater. However, as can be seen in this video, some algae grow faster than the fish can eat them. As one algae type is reduced, another takes over. The algae are out of control.
The best way to control algae is to have a good water balance.
You can look up various algae types on the net and what causes them - usually lack of this, too much of that etc - in short, the water is not in balance.
One way to control algae is to have fish or shrimps eating them. The Siamensis Flying Fox is an excellent algae eater. However, as can be seen in this video, some algae grow faster than the fish can eat them. As one algae type is reduced, another takes over. The algae are out of control.
The best way to control algae is to have a good water balance.
You can look up various algae types on the net and what causes them - usually lack of this, too much of that etc - in short, the water is not in balance.
- Let the plants and the substrate do the work. Over time, the plants will grow strong roots and leafs, and the substrate will develop a good bacteria culture.
- Use a gravel cleaner. The bottom in an aquarium is not endless. It is necessary to remove excess waste products from the gravel.
- Make frequent and large water changes. There is probably much too much of something, and making one large water change only removes the part, that is dissolved in the water. There is probably a lot more in the substrate, which will be released into the new water. Therefore a water change of 50% of the water twice a week is not overdoing it, when there is an algae attack.
- Use reverse osmosis purified water. If there's too much of something, then it may already be present in the tap water. Clean the water completely using reverse osmosis (RO). This effectively guarantees that there is no unknown surplus of anything in the water.
- Have good water circulation. It is very important, that there is a good water flow over the substrate, and there are no pockets of uncirculated water. Good water circulation combined with frequent water changes effectively cleans all corners in the aquarium.
- Remove any dead and decomposing plant leafs, as they only add to the algae growth.
- Remove as many algae by hand as you possibly can. You may have to sacrifice some healthy plant leafs if they are attacked by algae. The plant will have to grow new leafs. An old tooth brush is an excellent tool for removing algae without disturbing the decoration too much.
- Reduce lighting to 6 hours. This is enough for the plants, the rest is just used for algae growth, which is not desired.
- Replace lighting hardware. Flourescent light tubes wear out in approximately one year. They still work fine, but the colour spectrum changes or even degrades over time. Eventually it is not good enough for the plants. Therefore, the photosyntetis can't function effectively even though it looks like there is plenty of light.
- Have CO2-injection, preferrably from a pressure bottle and a diffuser/reactor. CO2 + light equals photosynthetis in the plant.
- Use fertilizer. It is tempting to think that fertilizer should be the cause of algae growth, but actually the fertilizer helps the plants to win over the algae in the fight for nutrients in the water. Do not over-dose, more is not better! Follow the instructions for the fertilizer product.
- Add fast-growing plants. Fast-growing plants will quickly use up all the excess nutrients. When they have done their job, they usually starts to deteriorate or just stop growing.
- Have algae-eating fish like Ancistrus and Siamensis Flying Fox.
- Have shrimps if you don't have shrimp-eating fish.
- DO NOT USE ANY POISON-PRODUCTS! Algae-killers will likely kill some of your plants too, maybe even some fish, and do nothing to improve the water balance. Cure the cause of the problem, not the symptom.
The above list is not in any particular order, all of the steps are important. The water changes need to be large in the beginning, but in a couple of weeks the water should be stable enough to allow for a more normal water change schedule. The other steps have to be followed until the algae are under control. But be careful about celebrating too early - a small and overlooked algae may wait to take over again!

Water chemistry
Water chemistry requires a visit to the chemistry lessons back in the school days. There is a big table of all the known atoms. Atoms combine into molecules, so while there can be an uncountable number of different molecules, there are only about one hundred basic atoms.
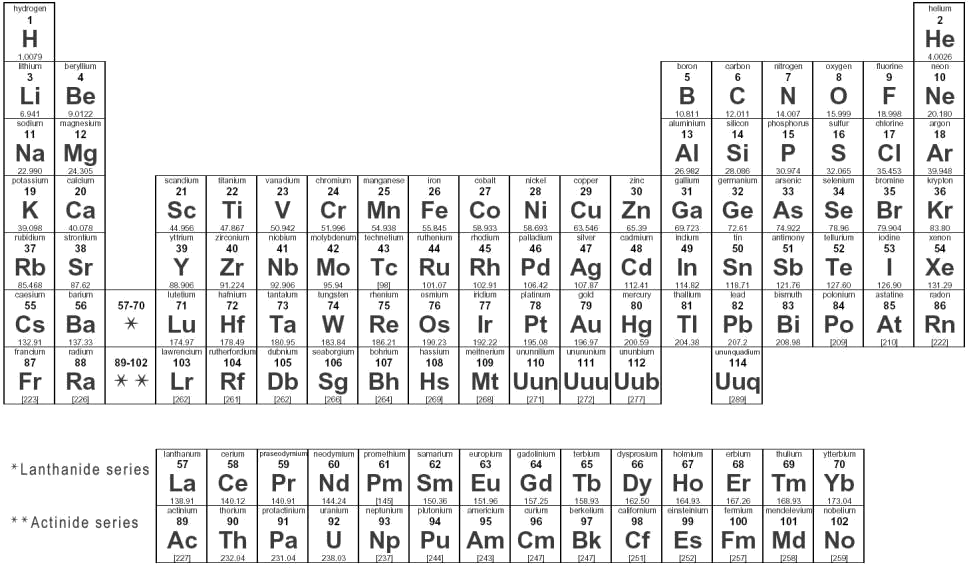
The periodic table is a list of all the known atoms in the world. If you read it horizontally, you will find Hydrogen (H) is number 1, Helium (He) is number 2 and so on. Carbon (C) is number 6, Nitrogen is number 7 and Oxygen (O) is number 8.
H2O is two hydrogon atoms and one oxygen atom combined into a water molecule.
CO2 is one carbon atom and two oxygen atoms combined into one carbon-dioxide molecule.
Photosyntethis is CO2 + light + plant activity = CO2 is split into carbon (C) for the plant and oxygen (O2) released into the water. You may want to add CO2 to your aquarium.
In the nature, there is a very fine balance of all of this, this balance is also needed in the aquarium.
The biological processes in the substrate will do a lot of the work, there is a cycle of waste products from fish and decomposing plants producing ammonia, this is then converted into nitrites and later nitrates. This process is essential for a good water balance.
Fertilizers are a selected mix from the periodic table of minerals and metals. There are no organic processes in fertilizer, it's only some extra elements, which help the plant to consume some of the other elements. The difference in fertilizer quality (and price) is how many of these extra elements are included. The difference between no fertilizer at all and a even just a cheap basic fertilizer is noticable.
NOTE: ONLY use special aquaraium fertilizer - NEVER use flower fertilizer which have a very different (and lethal for your fish) combination and concentration of elements.
N, P, K (Nitrogen #7, Phosphorous #15, Potassium #19) are the three primary elements for plant growth, and are known as macro nutrients.
All the rest are known as micro nutrients or trace elements.
These are typically Ca (Calcium), Mg (Magnesium), S (Sulphur), Fe (Iron), B (Boron), Zn (Zinc), Cu (Copper), Mn (Manganese), Mo (Molybdenum).
N (Nitrogen) is used by the plant for producing a lot of the proteins and amino acids needed during growth, and lack of it can be seen by the plant turning yellow and not growing. N is the most important element for the plant, and it is easy to run out in an aquarium.
P (Phosphorous) is essential for the photosynthetis process. It is only required in low quantities.
K (Potassium) is just like Nitrogen needed to build up the plant, and is also easily used up in an aquarium.
It is very difficult to precisely measure the amount of the various elements in the water, but over time the plants will tell how they are doing.
H2O is two hydrogon atoms and one oxygen atom combined into a water molecule.
CO2 is one carbon atom and two oxygen atoms combined into one carbon-dioxide molecule.
Photosyntethis is CO2 + light + plant activity = CO2 is split into carbon (C) for the plant and oxygen (O2) released into the water. You may want to add CO2 to your aquarium.
In the nature, there is a very fine balance of all of this, this balance is also needed in the aquarium.
The biological processes in the substrate will do a lot of the work, there is a cycle of waste products from fish and decomposing plants producing ammonia, this is then converted into nitrites and later nitrates. This process is essential for a good water balance.
Fertilizers are a selected mix from the periodic table of minerals and metals. There are no organic processes in fertilizer, it's only some extra elements, which help the plant to consume some of the other elements. The difference in fertilizer quality (and price) is how many of these extra elements are included. The difference between no fertilizer at all and a even just a cheap basic fertilizer is noticable.
NOTE: ONLY use special aquaraium fertilizer - NEVER use flower fertilizer which have a very different (and lethal for your fish) combination and concentration of elements.
N, P, K (Nitrogen #7, Phosphorous #15, Potassium #19) are the three primary elements for plant growth, and are known as macro nutrients.
All the rest are known as micro nutrients or trace elements.
These are typically Ca (Calcium), Mg (Magnesium), S (Sulphur), Fe (Iron), B (Boron), Zn (Zinc), Cu (Copper), Mn (Manganese), Mo (Molybdenum).
N (Nitrogen) is used by the plant for producing a lot of the proteins and amino acids needed during growth, and lack of it can be seen by the plant turning yellow and not growing. N is the most important element for the plant, and it is easy to run out in an aquarium.
P (Phosphorous) is essential for the photosynthetis process. It is only required in low quantities.
K (Potassium) is just like Nitrogen needed to build up the plant, and is also easily used up in an aquarium.
It is very difficult to precisely measure the amount of the various elements in the water, but over time the plants will tell how they are doing.
Bacteria and nitrification
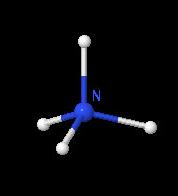
Nitrogen is a very important substance, and it is a part of many molecules (all amino-acids and subsequently DNA). Nitrogen N is number 7 in the periodic table. In the picture to the right, nitrogen is the blue atom in the middle, and the four grey atoms are hydrogen, making it a NH4 (ammonium) molecule.
Ammonium
Ammonia (NH3) and ammonium (NH4) comes from decomposing plant leafs and fish droppings. It is produced by bacteria in the decomposing process.
Nitrites and nitrates
Nitrites (NO2) and nitrates (NO3) are also produced by bacteria. Ammonium is converted to nitrites, and nitrites are converted into nitrates. The nitrates are needed by the plants.
Oxygen is needed for the process
The processes NH4 => NO2 => NO3 consume oxygen. If there is too much material to decompose (uneaten food, plant leafs, fish droppings, even dead fish), then the decomposing process may use up all the available oxygen and produce dangerously large amounts of ammonium at the same time. This can kill all the fish, and is a very real threat if the water is not in balance.
NH4 (ammonium) is dangerous in too large amounts.
NO2 (nitrite) is dangerous in too large amounts.
NO3 (nitrate) is not dangerous for the fish or for the pH level.
Frequent water changes remove the excess amounts, and a healthy bacteria culture in the filter and substrate converts all the dangerous ammonium and nitrites to nitrates for the plants.
Ammonium
Ammonia (NH3) and ammonium (NH4) comes from decomposing plant leafs and fish droppings. It is produced by bacteria in the decomposing process.
Nitrites and nitrates
Nitrites (NO2) and nitrates (NO3) are also produced by bacteria. Ammonium is converted to nitrites, and nitrites are converted into nitrates. The nitrates are needed by the plants.
Oxygen is needed for the process
The processes NH4 => NO2 => NO3 consume oxygen. If there is too much material to decompose (uneaten food, plant leafs, fish droppings, even dead fish), then the decomposing process may use up all the available oxygen and produce dangerously large amounts of ammonium at the same time. This can kill all the fish, and is a very real threat if the water is not in balance.
NH4 (ammonium) is dangerous in too large amounts.
NO2 (nitrite) is dangerous in too large amounts.
NO3 (nitrate) is not dangerous for the fish or for the pH level.
Frequent water changes remove the excess amounts, and a healthy bacteria culture in the filter and substrate converts all the dangerous ammonium and nitrites to nitrates for the plants.
Softness, hardness and pH
H2O without anything else is water as clean as it can get. It is also as soft, at it can get. You will normally read about fish from rivers, that they prefer soft water with a slightly low pH value. The water in these rivers is soft because it originates from melting snow in mountains and rain water. Water from the city water supply is usually hard, meaning that it contains some elements from the periodic table (calcium and magnesium in some form), that are not present in the river water.
It is made like that for a reason. A high CO2-concentration in soft water will lower the pH much more, than the same CO2-concentration in hard water would do. In other words, the hard water is more stable than soft water.
As you want to add CO2 to your aquarium, you will appreciate that the hard water can contain a high CO2-concentration without endangering the fish or plants.
The pH is a number which is neutral when around 7, acidic when lower (minimum value is zero) and alkaline when higher (maximum value is 14). If the pH changes too far away from neutral, then it becomes dangerous for living organisms like fish, plants and even human beings.
The nature of the pH number has to do with how hydrogen behaves in detail. It turns out, that H2O is a very simplified model of water. The two hydrogen atoms and one oxygen atom can react in other ways than forming the simple water molecule. Two water molecules ( 2 x H2O ) can convert into hydroxid (OH-) and hydronium (H3O+) - you can count the number of H's and O's and see, that it adds up. One hydrogen atom has moved from one of the water molecules leaving hydroxid (H2O minus H = HO or OH) to another water molecule forming hydronium (H2O plus H = H3O). It leaves its electron behind, so the hydroxid is OH- while the hydronium is H3O+.
The pH number is related to the activity of the hydroxid and hydronium generation. The behaviour of these (and other) variants of the water molecule is very complex, more information can be found in this wikipedia article about hydronium, hydroxid and pH or the youtube videos below. It's a long list of chemistry videos, I fast-forwarded to the video covering hydronium, hydroxid and pH.
It is made like that for a reason. A high CO2-concentration in soft water will lower the pH much more, than the same CO2-concentration in hard water would do. In other words, the hard water is more stable than soft water.
As you want to add CO2 to your aquarium, you will appreciate that the hard water can contain a high CO2-concentration without endangering the fish or plants.
The pH is a number which is neutral when around 7, acidic when lower (minimum value is zero) and alkaline when higher (maximum value is 14). If the pH changes too far away from neutral, then it becomes dangerous for living organisms like fish, plants and even human beings.
The nature of the pH number has to do with how hydrogen behaves in detail. It turns out, that H2O is a very simplified model of water. The two hydrogen atoms and one oxygen atom can react in other ways than forming the simple water molecule. Two water molecules ( 2 x H2O ) can convert into hydroxid (OH-) and hydronium (H3O+) - you can count the number of H's and O's and see, that it adds up. One hydrogen atom has moved from one of the water molecules leaving hydroxid (H2O minus H = HO or OH) to another water molecule forming hydronium (H2O plus H = H3O). It leaves its electron behind, so the hydroxid is OH- while the hydronium is H3O+.
The pH number is related to the activity of the hydroxid and hydronium generation. The behaviour of these (and other) variants of the water molecule is very complex, more information can be found in this wikipedia article about hydronium, hydroxid and pH or the youtube videos below. It's a long list of chemistry videos, I fast-forwarded to the video covering hydronium, hydroxid and pH.
Plants compete against each other
The plants all try to get what they need of N, P, K and other elements from the periodic table. Fast-growing plants consume more than slow-growing plants, and are thus good at removing excess nutrients from the water. But when these excess nutrients have been used up and things are normal again, then they starve and must be either removed or fed with fertilizer.
Slow-growing plants may have spent a lot of time setting up a large network of plant roots, which can effectively drain a large area for nutrients. If there is enough, then they grow larger until they have used it all. If there isn't enough, they just grow much slower.
Some plants won't even survive unless CO2 + special fertilizer is added and the substrate is of high quality.
Slow-growing plants may have spent a lot of time setting up a large network of plant roots, which can effectively drain a large area for nutrients. If there is enough, then they grow larger until they have used it all. If there isn't enough, they just grow much slower.
Some plants won't even survive unless CO2 + special fertilizer is added and the substrate is of high quality.
